Polyamide adhesion to dissimilar materials for structural
bonding
What are Nylon and Kevlar?
Nylon, Kevlar and Nomex are different forms of polyamide, the generic name for all fiber-forming polyamides with recurring amide groups (-CONH2).
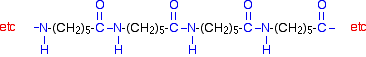
Polyamides comprise the largest family of engineering plastics with a very wide range of applications. Polyamides (nylons and Kevlar) are often formed into fibers and are used for monofilaments and yarns. Characteristically polyamides are very resistant to wear and abrasion, have good mechanical properties even at elevated temperatures, have low permeability to gases and have good chemical resistance, toughness, impact resistance and dimensional stability. The mythical stories of the etymology of the nylon name are rather humorous. Interestingly DuPont never created a trade mark for nylon.
Two of the most commonly known polyamides are Nylon-6 is known for its elasticity, dyability and recyclability while Nylon 6.6 is known for its heat resistance, rigidity, dimensional stability and abrasion resistance.
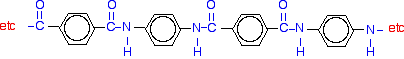
Kevlar® and Nomex®, produced by DuPont are members of the aramid family of nylons. They are similar in structure to nylon-6,6 except that instead of the amide links joining chains of say, six carbon atoms together (-6- hence its name), they join benzene rings. Kevlar is a very strong material - about five times as strong as steel, weight for weight. Kevlar is used for high performance racing tires, racing bike tires, cables and rope, brake linings, fabric for sails and parachutes, and to make composites for boat hulls and and body armor.
Go to links NYLON and KEVLAR for a more detailed description.
What are some uses of Polyamide and Aramid polymers?
The number of applications for nylon is enormous; a few examples include tie wraps, fabric hook-and-loop fasteners (e.g. trade name Velcro tape), electrical connectors, tubing, electrical housings, fishing line, automotive oil pans, many military applications, and a multitude of fabrics and carpeting.
Commercially nylon is commonly used in the production of tire cords, rope, belts, filter cloths, sports equipment and bristles. It is particularly useful when machined into bearings, gears, rollers and thread guides. These applications take advantage of the toughness and slippery nature of nylon. Polymers used in similar applications are UHMW, fluoropolymers such as PTFE and FEP (both are known as Teflon) and acetal (Delrin), which are much more expensive.
Kevlar fiber is used in bulletproof vests, as is UHMW PE fiber, in composites for boat construction, in lightweight mountaineering ropes, strength members in electrical cables, and for lightweight skis and racquets - amongst many other things.
One major commercial electronic instrumentation manufacturer successfully replaced the commonly used older technology of Chalkfast orange epoxy with BONDiTTM B-482 to terminate Kevlar strength members of large electrical cables, resulting in superior performance and termination strength that exceeded the cable and Kevlar rope strength.
How do you bond Nylon and Kevlar with an adhesive to dissimilar materials?
Nylon generally is considered bondable. Nylon can be bonded to itself and to other substrates without significant preparations (light abrasion is usually recommended) using BONDiTTM brand adhesives manufactured by RELTEK.
At room temperature, bonding using conventional solvents will not provide effective bonds on nylon. Some non-conventional solvent cements are sometimes used. Nylon dissolved in formic acid forms a high strength bond, but the cement is very toxic and highly corrosive, which makes it unfavorable for most modern manufacturing environments. A solution of resorcinol or calcium chloride and ethanol offers a less toxic formulation--the bonds are best when cured with heat. The other major issue is the solvent adhesives tend to form brittle joints, lack toughness, and are not suitable for dissimilar material bonding. Current trends for environmental safety have led industry away from solvents.
Nylon parts can be joined by various thermal welding techniques including vibration, ultrasonic, and spin welding. Welded joints of thermoplastics typically are subject to stress cracking after a period of use.
Because of these perceived limitations, generally for most applications conventional adhesives are used. The most common adhesives for nylon-to-nylon bonding are based on epoxy and phenol-formaldehyde formulations, although most adhesives are considered weak due to their creating a brittle joint. Hence, semi-rigid or flexible adhesives are preferred with good toughness in formation of an adhesive joint.
Adhesives often recommended include nylon-phenolic, nitrile-phenolic, nitriles, neoprene, modified epoxy, cyanoacrylate, modified phenolic, resorcinol-formaldehyde, and polyurethane. Bonds in the range of 250-1000 PSI (1.7-6.9 MPa) depending on the thickness of the adherends, have been obtained. [Arthur Landrock, Adhesives Technology Handbook, 1985, p. 195]. Caution is required with utilizing any adhesive containing formaldehyde which is a known carcinogen.
Additional considerations also make nylon adhesion more difficult. One is their propensity for high moisture pick-up, which makes careful drying imperative. Nylon will typically absorb 1 - 2 % moisture when left in air, and up to 8% when submerged 24 hours. Moisture has the effect of plasticizing the nylon so absorbed moisture could accelerate any migration of additives from the bulk to the surface of the film affecting adhesion or surface wetting tension.
With light abrasion some of the paired hydrogen and polar carboxyl groups can be broken up to create tertiary hydrogen (connected to a tertiary carbon and not sterically hindered or paired with another adjacent hydrogen) which are chemically active, along with the reactive carboxyl groups, and increase the surface energy. BONDiT B-4x series epoxy products are designed to take advantage of the tertiary hydrogen and polar sites to form good bonds.
Among the other substrates which nylon can be bonded to using BONDiTTM products
are: Delrin, UHMW, fluoropolymers, rubber, polyethylene, polyolefin, polypropylene, polyurethane, silicone, thermoplastics, thermoset, glass and ceramics, wood, metals, stone, aggregate and concrete.
Corona (plasma) or flame etch is occasionally used to prepare the nylon surface which
strengthens the bond by reacting surface chains with oxygen to form oxane bonds, significantly increasing the bond strength. BONDiTTM B-4x series epoxies readily form high strength bonds to the oxide rich substrate. Chemical etching with RELTEK Enchant has also been successfully employed to increase the surface energy of the nylon.
Most application do not need such enhancement and work well with just the
B-45TH adhesive on a lightly abraded (#100-120 grit) surface. The need to pretreat
the nylon by etching occurs in about 15% of the applications. If abrasion is not possible the BONDiTTM B-46TH has the capability of adhering to the unabraded nylon surface. It will even bond similarly to untreated Teflon and Delrin.
If you think that your application may require surface treatment, please call
us
at (707) 284-8808 or email us at reltek@reltekllc.com for technical support as this technique
requires some skill and experience. We can provide you with written instructions to guide you through a process for which in our experience there is a high degree of miss information extant within industry.
Fillers of nylon, Kevlar and Nomex may affect bonding by enhancement or degradation of adhesion. Typically mineral based fillers such as glass fiber tend to significantly increase adhesion when using RELTEK BONDiTTM products.
On the other hand, plasticizer additives inherently are designed to break or hinder crosslinking of plastic matrices (networks of hydrocarbon polymer resin.) They also will bloom to the surface over a period of time, and such activity is accelerated by heat. Hence, the phenomenon of a strong bond failing over time due to migration of the plasticizer into the adhesive joint. High level of plasticizer such as used with flexible PVC will greatly hinder any adhesion at all. Special techniques are employed to overcome this condition. One of those is the use of BONDiT A-3 in multiple coats as a primer to seal off the plasticizer from the adhesive joint.
For Kevlar surface treatment is not required. Aramid polymers form strong pi-stacking hydrogen bonding, as the source of their extraordinary steel-like strength. It is believed that the B-4X series epoxies interfere with the carbonyl (oxygen) bonding to the adjacent hydrogen on the phenyl groups. This opens up the carbonyl oxygen to form a covalent bond. Hence, BONDiTTM B-482 can form a bond that is stronger than the Kevlar fiber.
How do you design with NYLON for Harsh Environments?
When designing for practical applications consider all nylons are susceptible to hydrolysis, especially by strong acids. The molecular weight of nylon products so attacked drops fast, and cracks form quickly at the affected zones. Lower members of the nylons (such as nylon 6) are affected more than higher members such as nylon 12. This means that nylon parts cannot be used in contact with sulphuric acid for example, such as the electrolyte used in lead-acid batteries.
Nylons are much more resistant to alkaline hydrolysis. Hydrolysis is faster at higher temperatures. Hydrolysis by water alone is slow, but considered a disadvantage with related dimensional instability and degradation of electrical properties.
The water absorption of nylons generally decreases as the length of the carbon chain increases: higher nylons (longer chains) absorb less water than the lower nylons. Also, the even-numbered nylons absorb less water than the odd-numbered nylons. Absorption of water results in a slight decrease in tensile strength, a major increase in ultimate elongation, and a corresponding decrease in flexural modulus.
[Irving Skeist, Handbook of Adhesives, 3rd Ed, p. 485].
When being molded, nylon must be dried to prevent hydrolysis in the molding machine barrel since water at high temperatures can also degrade the polymer. Other molding design concerns are high shrinkage and high notch sensitivity.
The absorbed water can create a weak boundary layer under certain conditions. Generally, parts are dried moisture before bonding. Caution must be observed with mating the nylon to another substrate. The nylon part will grow and shrink due to the increase and egress of moisture from within the substrate. This will lead to stresses at the interface that may cause warpage and degradation of the bond strength. [Edward Petrie, Handbook of Adhesives and Sealants, 2000, p. 637]. The tough and flexible BONDiTTM B-45TH and B-482TH will handle the stresses from swelling and shrinkage, providing release and redistribution of the dislocation forces.
On the other hand, Kevlar is rather more resistant to hydrolysis than nylon. Shrinkage of Kevlar 29 and 49 (DuPont) in boiling water is less than 0.1%. Moisture content of Kevlar 29 at ambient absorption in air is about 7.0%, and "Bone-Dry Yarn" of 3.5%. Kevlar 49 by comparison is the same moisture content in air and dry of 3.5%.
BONDiTproducts are among the few available that as a polyamide adhesive will bond
nylon to itself or other substrates with differing coefficients of
thermal expansion
(CTE). BONDiTTM products are renowned
for their resistance to harsh environments and for
withstanding
long
term
deployment without debonding.
Bonding nylon to dissimilar materials is a particular problem due to it's high thermal expansion coefficient--twelve times that of steel, and among the higher of the engineering plastics--twice that of acetal and about the same as polypropylene. As a consequence the adhesive bond joint must be designed to accommodate the expansion/contraction differential between materials to avoid bond failure. The BONDiTTM B-4X series epoxies are especially designed for that purpose, ranging in elongation (stretch) from 10% to 500%. Adhesive joints designed with these products have been successfully thermal cycled -50°F to+150°F, and even cryogenic temperatures (liquid nitrogen) and as high as 300°F. Nylon is suitable for cryogenic applications as is PPS. BONDiTTM B-4X series epoxies are an excellent choice for bonding to dissimilar materials such as Nd-Fe-B magnets.
In addition, BONDiT B-4X epoxies are not normally hindered by the presence of moisture at the bond line. So nylon which is not fully dry can still be bonded.
BONDiTTM B-45TH produces an excellent bond with nylon (amide) and Kevlar and Nomex (aramid) fabrics. With its inherent high flexibility the bond joint retains some flexibility. However, through bonding the parallel fibers together the fabric will naturally stiffen because the fibers can no longer slide past each other which is what gives fabric its flexibility. The B-45TH and B-482TH can be used to bond Velcro® to many substrates. These adhesives provide toughness, non-brittleness, chemical resistance such as exposure to chemical (dry) cleaning, hot water washing and drying cycles, and steam, including autoclaving.
Our first product recommendation for bonding nylon is BONDiTTM B-45TH or BONDiTTM B-482TH.
Our recommendation would change if you are overmolding or if there are other
special adhesive needs.
If overmolding, we would recommend priming the substrate
with
our adhesion promoters BONDiTTM A-43, A-3, C-6, C-52, or A-46. An example would be molding thermoplastic polyurethane, two-part urethane rubber or rubbers in general such as EPDM to nylon.
Or, if exceptional chemical resistance
is
desired,
we would recommend the BONDiTTM B-481 or B-4811.
Abundant engineering support is provided at RELTEK for our customers
Please feel free to call or email us with your questions. Our recommendations are founded in compressive experience. We are willing to share that experience with you to ensure your success—whether or not you become a RELTEK customer, we welcome the opportunity to serve you.
Our promise
While solving the insolvable
With extraordinary customer service
We stick to you forever